F18 PET scans test for the amyloid plaques that are the culprits behind Alzheimer’s.
The Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging (SNMMI) assembled an Amyloid Imaging Taskforce (AIT) consisting of dementia and imaging experts to review the scientific literature and develop consensus recommendations for the clinical use of this technology.
The AIT concluded that amyloid imaging could potentially be helpful in the diagnosis of people with cognitive impairment when considered along with other clinical information, and when performed according to standardized protocols by trained staff. In addition, they emphasized that the decision whether or not to order amyloid imaging should be made only after a comprehensive evaluation by a physician experienced in the assessment and diagnosis of cognitive impairment and dementia, and only if the presence or absence of amyloid would increase certainty in the diagnosis and alter the treatment plan.
Who Should Use F18
According to the AIT, appropriate candidates for amyloid PET imaging include:
- Those who complain of persistent or progressive unexplained memory problems or confusion and who demonstrate impairments using standard tests of cognition and memory.
- Individuals meeting tests for possible Alzheimer’s, but who are unusual in their clinical presentation.
- Individuals with progressive dementia and atypically early age of onset (before age 65).
What are F18 Scans & What do they Show?
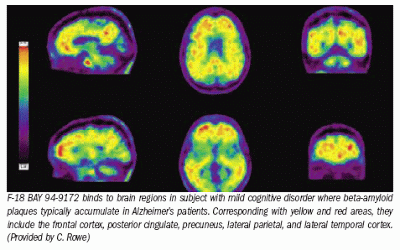
F18 PET-scans use radiopharmaceuticals (radioactive drugs) to produce three-dimensional functional images of the brain. In taking F18 scans to see amyloid in the brain, the radiopharmaceutical is introduced into the body by injection into a vein and binds specifically to the amyloid protein. This enables visualization of areas in the brain where amyloid has clumped together into plaques. One of the F18 PET-scan compounds, approved by the U.S. Food and Drug Administration, is called Amyvid florbetapir.
When using an F18 PET-scan to look into the brain of a person with dementia, doctors can get clarify some critical issues.
- If the F18 scan shows no amyloid buildup in their brain, then the cause of the dementia is very likely to be something other than Alzheimer’s disease. Other causes of dementia include: strokes, thyroid problems, drug interactions, chronic alcoholism, and vitamin deficiencies.
- If an F18 scan shows that a person with memory impairment has amyloid buildup in their brain, this increases the likelihood that the memory impairment is caused by Alzheimer’s disease, but it remains a likelihood, not a certainty.
- If a person without memory complaints or impairment takes a scan and shows amyloid buildup in the brain, it does not necessarily mean that they will develop Alzheimer’s. Many people have amyloid in their brains but are cognitively normal. More research is needed to understand the significance of amyloid plaques in this population.
When Should F18 Be Avoided
Inappropriate candidates for amyloid PET imaging include:
- Those who are age 65 or older and meet standard definitions and tests for Alzheimer’s, since a positive PET scan would provide little added value.
- Asymptomatic people or those with a cognitive complaint but no clinical confirmation of impairment.
Amyloid PET imaging is also inappropriate:
- As a means of determining the severity of dementia.
- When requested solely based on a family history of dementia or presence of other risk factors for Alzheimer’s, such as the ApoE-e4 gene.
- As a substitute for genetic testing for mutations that cause Alzheimer’s.
- For non-medical reasons, such as insurance, legal or employment decisions.
“As amyloid imaging becomes more prevalent in clinical settings, medical professionals must understand how to appropriately utilize the test,” said Frederic H. Fahey, D.Sc., SNMMI president. “Neurology and dementia experts should order the test only when appropriately indicated, and nuclear medicine and molecular imaging professionals must ensure they have been adequately trained to interpret the results of the scan. Working together, we hope that the information garnered from amyloid PET imaging will aid in diagnosis and play a pivotal role in the development of new treatments for Alzheimer’s.”
How Should F18 Be Used
The taskforce acknowledged that the healthcare provider makes the ultimate judgment regarding the care of each patient. The AIT sought to assist this process and identified the following general sequence of events for the use of an F18 amyloid PET scan according to the new criteria:
- Evaluation by a dementia expert to assess the need for diagnostic testing, possibly to include amyloid PET if the AUC are met.
- Referral to a qualified provider of amyloid PET services.
- Performance, interpretation and reporting of the amyloid PET scan according to established standards.
- Incorporation of the PET result into the clinical assessment process.
- Disclosure of the PET result by the clinician to the patient and caregivers, along with discussion of the result and its management consequences.
Although identifying potential benefits, the AIT concluded that amyloid PET results will not constitute and is not equivalent to a clinical diagnosis of Alzheimer’s disease dementia. They said that imaging is only one tool among many that clinicians should use judiciously to manage patients, and that amyloid PET imaging does not substitute for a careful history and examination.
“Because both dementia care and amyloid PET technology are in active development, these new appropriate use criteria will require periodic reassessment and updating,” Carrillo said.
Cost
Amyloid imaging is not covered by insurance at this time, and costs for the scan are “out of pocket.” While costs of amyloid PET are not yet established, and PET costs in general can vary depending upon location, other PET scans are known to cost between $1,000 and $3,000, or more. Nonetheless, the AIT concluded that the proven sensitivity and specificity of the new radiopharmaceuticals for brain amyloid and the known association between brain beta amyloid deposition and Alzheimer’s suggest these new radiopharmaceuticals could potentially be helpful in the workup and diagnosis of patients with cognitive impairment.
Medicare
Leading experts have recommended guidelines to Medicare stating that amyloid imaging could potentially be helpful in the diagnosis of people with cognitive impairment when considered along with other clinical information, and when performed according to standardized protocols by trained staff. The guidelines also indicate that, although published data is extremely limited, amyloid PET is likely to contribute to better patient care under specific circumstances.
MORE INFORMATION
About the Society of Nuclear Medicine and Molecular Imaging
The Society of Nuclear Medicine and Molecular Imaging (SNMMI) is an international scientific and medical organization dedicated to raising public awareness about nuclear medicine and molecular imaging, a vital element of today’s medical practice that adds an additional dimension to diagnosis, changing the way common and devastating diseases are understood and treated and helping provide patients with the best health care possible. SNMMI’s more than 19,000 members set the standard for molecular imaging and nuclear medicine practice by creating guidelines, sharing information through journals and meetings and leading advocacy on key issues that affect molecular imaging and therapy research and practice. For more information, visit www.snmmi.org.
About the Alzheimer’s Association
The Alzheimer’s Association is the world’s leading voluntary health organization in Alzheimer care, support and research. Our mission is to eliminate Alzheimer’s disease through the advancement of research, to provide and enhance care and support for all affected, and to reduce the risk of dementia through the promotion of brain health. Our vision is a world without Alzheimer’s. Visit www.alz.org or call 800-272-3900.
SOURCES:
- Eli Lilly & Co.
- Alzheimer’s Association
- Fluorine-18 Radioactive Tracers