VIDEO + ARTICLE:
The Stable Isotope Labeling Kinetics (SILK™) blood test is a recent technology that sees Alzheimer’s years before signs appear. It can open doors to researchers trying to solve problems like the connection between Alzheimer’s and aging. See how.
Biomarkers in the blood, indicating Alzheimer’s, may appear 15 years before there are signs of trouble. Researchers are using the SILK™ blood test to track those biomarkers and gain a better shot at finding ways to prevent and treat Alzheimer’s.
The Stable Isotope Labeling Kinetics (SILK™) platform was pioneered at Washington University School of Medicine (WUSM) in St. Louis and is marketed by C2N. It includes new technologies that enable a novel approach to measure the metabolism of brain-derived proteins implicated in Alzheimer’s disease (AD) and mild cognitive impairment (MCI).
Continued below video…
Why Does Aging Increase Alzheimer’s?
The greatest risk factor for Alzheimer’s disease is advancing age. After 65, the risk doubles every five years. As many as half of the elderly who are over 85 are estimated to be living with some type of dementia, usually Alzheimer’s.
A sizable study demonstrated that in our 30s, a healthy brain clears amyloid-beta every four hours. At 80 years old, it takes more than 10 hours. This may
explain the link between age and Alzheimer’s, as well as what we need to do to fight this disease.
Researchers at Washington University School of Medicine in St. Louis have identified some of the key changes in the aging brain that lead to the increased risk. The changes center on amyloid beta 42, a main ingredient of Alzheimer’s brain plaques. The protein, a natural byproduct of brain activity, normally is cleared from the brain before it can clump together into plaques. Scientists long have suspected it is a primary driver of the disease.
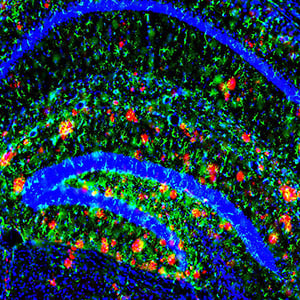
A new study reveals that the brain’s ability to clear the main ingredient of Alzheimer’s plaques slows with age (the plaques are red in this image). The findings could help explain why risk of the disease increases with age.
“We found that people in their 30s typically take about four hours to clear half the amyloid beta 42 from the brain,” said senior author Randall J. Bateman, MD, the Charles F. and Joanne Knight Distinguished Professor of Neurology. “In this new study, we show that at over 80 years old, it takes more than 10 hours.”
The slowdown in clearance results in rising levels of amyloid beta 42 in the brain. Higher levels of the protein increase the chances that it will clump together to form Alzheimer’s plaques.
The result details appear online in the Annals of Neurology.
Results Sum-Up
For the study, the researchers tested 100 volunteers ages 60 to 87. Half had clinical signs of Alzheimer’s disease, such as memory problems. Plaques had begun to form in the brains of 62 participants.
The subjects were given detailed mental and physical evaluations, including brain scans to check for the presence of plaques. The researchers also studied participants’ cerebrospinal fluids using a technology developed by Bateman and co-author David Holtzman, MD, the Andrew B. and Gretchen P. Jones Professor and head of the Department of Neurology at Washington University. The technology — known as stable isotope-linked kinetics (SILK)— allowed the researchers to monitor the body’s production and clearance of amyloid beta 42 and other proteins.
In patients with evidence of plaques, the researchers observed that amyloid beta 42 appears to be more likely to drop out of the fluid that bathes the brain and clump together into plaques. Reduced clearance rates of amyloid beta 42, such as those seen in older participants, were associated with clinical symptoms of Alzheimer’s disease, such as memory loss, dementia and personality changes.
Scientists believe the brain disposes of amyloid beta in four ways: by moving it into the spine, pushing it across the blood-brain barrier, breaking it down or absorbing it with other proteins, or depositing it into plaques.
“Through additional studies like this, we’re hoping to identify which of the first three channels for amyloid beta disposal are slowing down as the brain ages,” Bateman said. “That may help us in our efforts to develop new treatments.”
How the SILK™ Blood Test Works
Biomarkers are chemicals in our bodies that are markers for an illness, such as Alzheimer’s. Until now, doctors looking for Alzheimer’s biomarkers would go through the delicate process of taking cerebrospinal fluid from patients. For the first time, instead of analyzing Alzheimer’s proteins in cerebrospinal fluid, the SILK™ Blood Test makes it possible to detect the same metabolic markers in patients’ blood samples.
This capability has implications for the advancement of new treatments, early prevention, and personal wellness. Alzheimer’s is now one of the major global healthcare concerns. Approximately 44 million people currently have clinical Alzheimer’s disease (AD). Millions more have MCI that places them at high risk for progression to clinical AD. The number of cases of AD and MCI are expected to increase sharply in the years ahead due to the aging baby boomer population.
Pharmaceutical companies developing new drugs targeting AD increasingly recognize that early intervention provides the greatest chance of halting or reversing disease progression. Biomarkers are needed to detect this early pathology, which can begin at least 15 years before the onset of any clinical symptoms. At the same time, dynamic biomarkers, like those offered by the SILK™ platform, may also track treatment responses during the pre-symptomatic stages of disease.
Since 2008, C2N has applied the SILK-Aβ® test to measure the kinetics of beta-amyloid in cerebrospinal fluid. The test has served as a primary endpoint in clinical drug studies to demonstrate target engagement and guide dose selection. The SILK-Aβ® isoforms test is also highly sensitive to identifying people with brain amyloidosis (one of the earliest indicators of Alzheimer’s), even before amyloid deposits are seen with brain imaging. Still, the more invasive nature of cerebrospinal fluid sampling has impeded the full potential of the SILK-Aβ® method.
“With a simplified SILK-Aβ® test available through blood sampling, we now have an opportunity to validate a unique therapeutic and diagnostic marker,” stated Dr. Joel B. Braunstein, CEO of C2N. “We plan to achieve this validation by collaborating with pharmaceutical companies that are testing their compounds in phase 2 and phase 3 clinical studies, as well as by participating in natural history studies tracking the progression of AD. If successful, we expect to be able to offer a reliable and informative screening test that is also convenient for patients.”
Alzheimer’s Disease & Mild Cognitive Impairment
Currently AD is diagnosed based on clinical assessment by skilled neurologists or based on an amyloid imaging brain scan: there are no approved blood based biomarkers for this disease. Early detection of AD is a major research focus since early detection would pave the way for early intervention. Mild cognitive impairment (MCI) is often a precursor of clinical AD. The disorder is associated with cognition changes that are serious enough to be noticed by the individuals experiencing them or to other people, but the changes are not severe enough to interfere with activities of daily living. Individuals with MCI have a significantly increased risk of eventually developing AD, with approximately 50% of such individuals converting to AD within three years.
SILK-Aβ® Technology
Dr. Randall Bateman and Dr. David Holtzman originally developed the SILK™ technology at WUSM. Scientific American recognized the SILK-Aβ® assay as one of the top 50 new innovative technologies of the year. Similar to a pulse chase assay, the SILK-Aβ® assay relies on in vivo labeling of human subjects with a stable isotope labeled amino acid. The stable isotope labeled amino acid is non-radioactive, safe to the environment and to humans, and is incorporated into newly generated proteins. Using a highly sensitive mass spectrometer, we can measure the incorporation of the stable isotope into Aβ and thereby assess the metabolism of Aβ. This ability is particularly useful for early detection of brain amyloidosis or when assessing the pharmacodynamic effect of drugs that are hypothesized to alter the metabolism of Aβ in humans. With these new developments, we have expanded the use of the assay to measuring Aβ metabolism in plasma.
MORE INFORMATION:
- SUPPORT: This research was supported by the National Institutes of Health (NIH), grants R01NS065667, K23AG030946, P50 AG05681, P01 AG03991, UL1 RR024992, P30 DK056341, P41 GM103422 and P30 DK020579; the Adler Foundation; and by philanthropic gifts from Edwin and Barbara Shifrin and Jeff Roschman.
- REFERENCE: Patterson BW, Elbert DL, Mawuenyega KG, Kasten T, Ovod V, Ma S, Xiong C, Chott R, Yarasheski K, Sigurdson W, Zhang L, Goate A, Benzinger T, Morris JC, Holtzman D, Bateman RJ. Age and amyloid effects on human central nervous system amyloid-beta kinetics. Annals of Neurology, online July 20, 2015.
-
About C2N Diagnostics
C2N Diagnostics, LLC (www.c2ndiagnostics.com) formed in late 2007 by scientific co-founders Drs. David Holtzman and Randall Bateman of Washington University School of Medicine in St. Louis, the Washington University Office of Technology Management, and LifeTech Research, a Maryland-based technology research and commercialization firm (www.lifetechresearch.com). C2N is commercializing a suite of novel biomarker assays and tools to assist in pre-clinical drug discovery, clinical drug development, and the early detection and assessment of progression of debilitating neurodegenerative disorders. The company’s products include the SILK-Aβ®, SILK-ApoE™, SISAQ-Aβ™, and SISAQ-Tau™ Assays, which rely upon stable isotope labeling and tandem mass spectrometry for the measurement of the kinetics, or in vivo metabolism, and absolute quantitation of brain derived proteins. Beyond AD, products are in development to target Parkinson’s disease, Huntington’s disease, brain injury, schizophrenia and amyotrophic lateral sclerosis, among other conditions.
SOURCE:
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient-care institutions in the nation, currently ranked sixth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.