For years, Alzheimer’s drug development has focused largely on one target: amyloid — the sticky protein that builds up in the brain long before symptoms appear.
Now, another major Alzheimer’s target may finally be showing signs of progress.
Biogen, makers of the successful Alzheimer’s drug Leqembi, recently announced topline results from a Phase 2 study of diranersen (BIIB080). Diranersen is an experimental drug designed to reduce tau, a different protein that many scientists believe is more closely tied to memory loss and worsening symptoms in Alzheimer’s disease.
The results were mixed.
The study missed its main goal — a disappointment that would normally cool enthusiasm.
Yet at the same time, the trial showed something researchers have been trying to demonstrate for years: signs that reducing tau may also translate into meaningful benefits for thinking and daily function.
That combination — setback plus scientific progress — is exactly why many experts are paying close attention.
What Is Diranersen?
Diranersen is not another version of Leqembi or Kisunla.
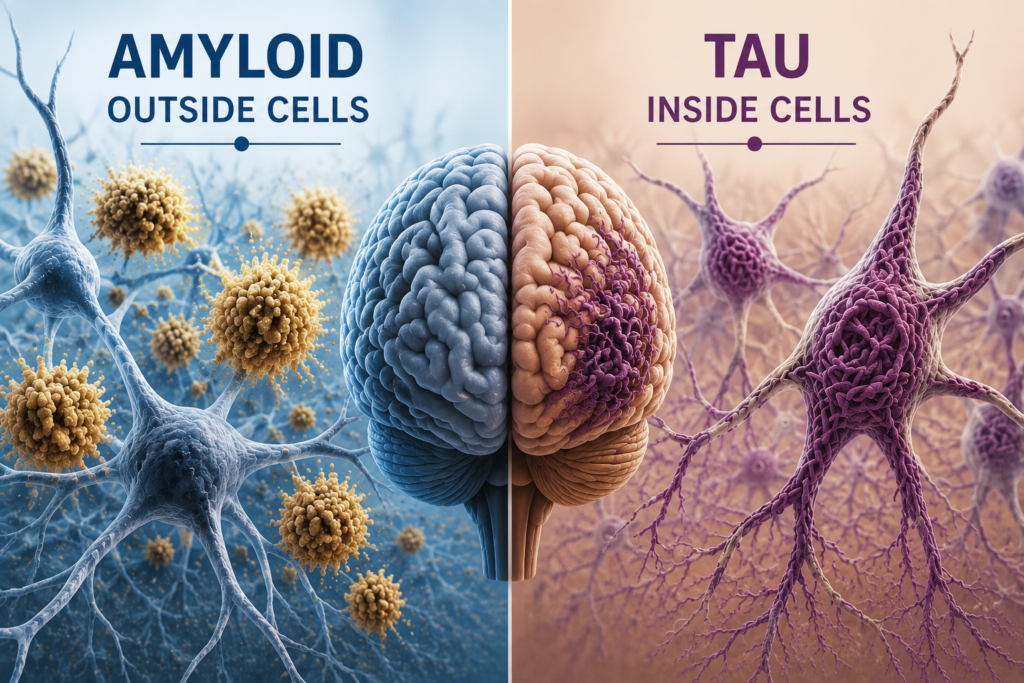
Those drugs target amyloid plaques in the brain. Diranersen targets tau, a protein that becomes twisted and tangled inside brain cells.
Many researchers now believe Alzheimer’s may work something like this:
- Amyloid helps start the disease process.
- Tau helps drive symptoms and progression.
In other words, amyloid may light the match, but tau may spread the fire.
That is one reason scientists have long hoped that targeting tau could produce stronger effects on cognition and day-to-day functioning.
The problem?
Until now, tau drugs have repeatedly disappointed.
What Happened in the Study?
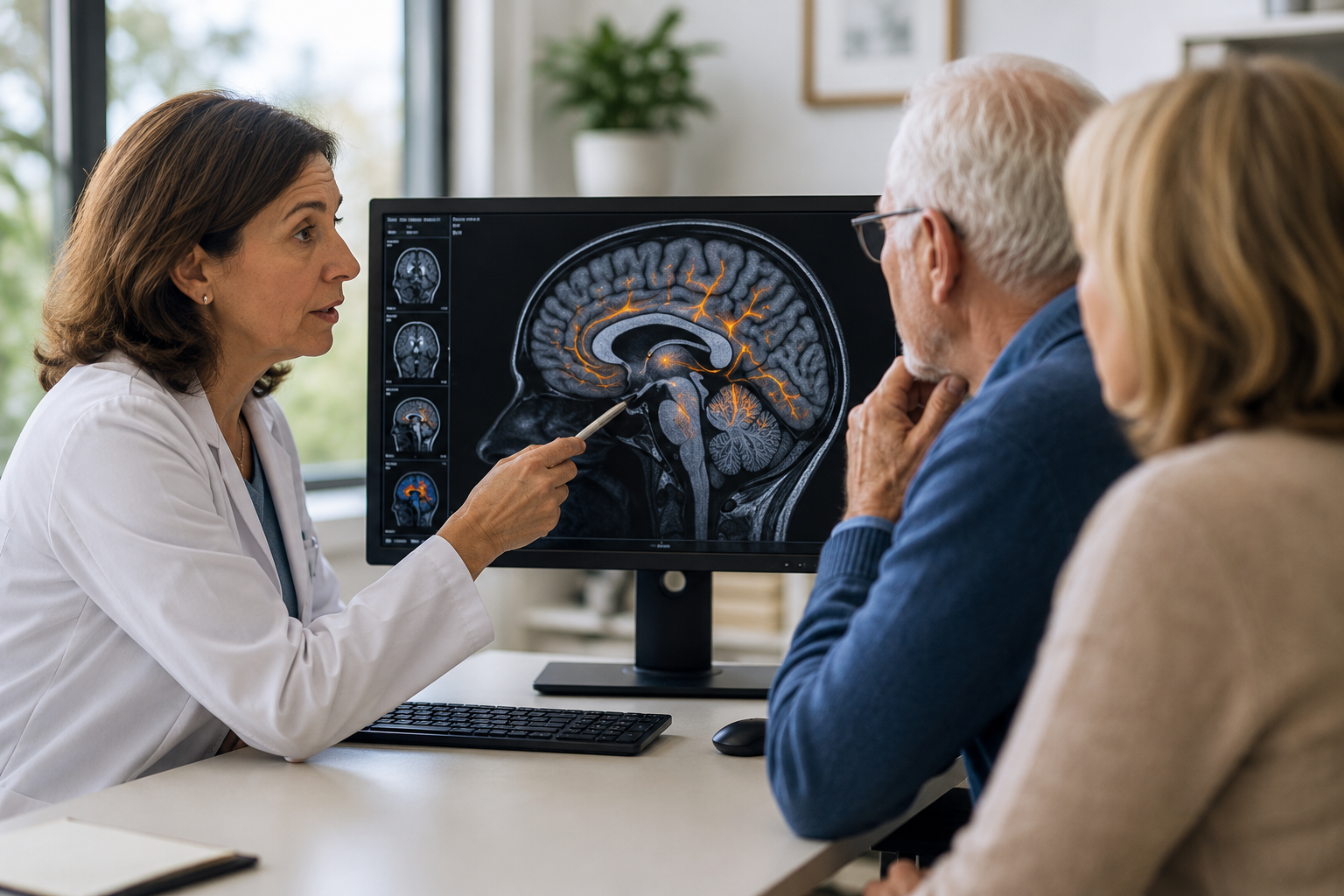
The Phase 2 CELIA study tested diranersen in people with early Alzheimer’s disease.
Biogen reported that the drug did not meet its primary endpoint, meaning it failed to show a statistically significant dose-response benefit on a commonly used Alzheimer’s measure called the Clinical Dementia Rating–Sum of Boxes (CDR-SB).
That matters because the primary endpoint is the study’s main pre-agreed test — the result regulators usually focus on most.
Normally, missing the primary endpoint would be viewed as a major problem.
But this study was more complicated.
Biogen also reported encouraging findings in several pre-specified secondary measures, including:
- Reduced tau biomarkers in spinal fluid
- Reduced tau accumulation on PET brain scans
- Signals suggesting slower cognitive decline, especially at the lower dose
Perhaps most notably, the company described this as the first study to show both reduction in tau pathology and accompanying clinical benefit signals.
That claim will likely receive intense scrutiny from scientists, but if confirmed in later trials, it could prove important.
Why the Nuance Matters
Caregivers and families have been through many waves of Alzheimer’s “breakthrough” headlines.
Sometimes the excitement proved justified.
Sometimes it did not.
That history matters.
A headline saying:
“Drug reduces tau and improves cognition”
sounds dramatic.
But without context, it risks creating expectations that later lead to disappointment.
The more accurate picture is this:
Diranersen did not clearly succeed — but it did not clearly fail either.
Instead, the study may have uncovered something scientifically important:
A possible signal that lowering tau could actually affect symptoms.
That is different from simply changing a biomarker.
For families, this distinction matters because the ultimate question is not:
“Did the scan improve?”
It is:
“Will this help someone think better, function longer, or decline more slowly?”
The encouraging part of the diranersen data is that researchers believe they may finally be seeing hints of that connection.
Why Tau Matters So Much
Tau has become one of the most watched areas in Alzheimer’s research.
Scientists increasingly suspect that tau may correlate more closely with:
- Memory loss
- Functional decline
- Disease severity
People can carry significant amyloid in the brain for years before symptoms appear.
Tau spread, however, often tracks more closely with worsening cognition.
If future studies confirm that reducing tau can slow decline, it could reshape Alzheimer’s treatment.
Instead of relying mostly on amyloid drugs, future treatment may involve combination therapy — perhaps an amyloid drug like Leqembi plus a tau-targeting therapy such as diranersen.
That possibility remains years away, but it helps explain why researchers are watching these results so carefully.
What This Means for Families Right Now
For now, nothing changes immediately.
Diranersen is not approved, was tested in early Alzheimer’s, and still requires larger studies to prove whether the benefits are real and meaningful.
Families should not view this as a treatment option available today.
But they also should not ignore it.
Even though the study hit a setback, many researchers see the results as an important proof-of-concept moment:
A signal that tau — one of Alzheimer’s most difficult targets — may finally be becoming druggable.
That may not mean faster progress tomorrow.
But it could mean the field just took a meaningful step toward the next generation of Alzheimer’s treatments.