A Competition That Isn’t Only About the Drug
When new Alzheimer’s treatments reach the market, the focus usually falls on the drug itself—how effectively it removes amyloid plaques, how much it slows cognitive decline, and how safe it is.
But for disease-modifying therapies like Leqembi, the medication is only part of the equation.
These treatments depend on a complex medical pathway that includes:
- Early diagnosis
- Biomarker confirmation
- Imaging monitoring
- Infusion-based treatment
This means the drug that fits best into the existing healthcare infrastructure may gain an important advantage.
Why Alzheimer’s Treatment Now Requires Biological Diagnosis
For decades, Alzheimer’s disease was diagnosed mainly through symptoms and cognitive testing.
That has changed.
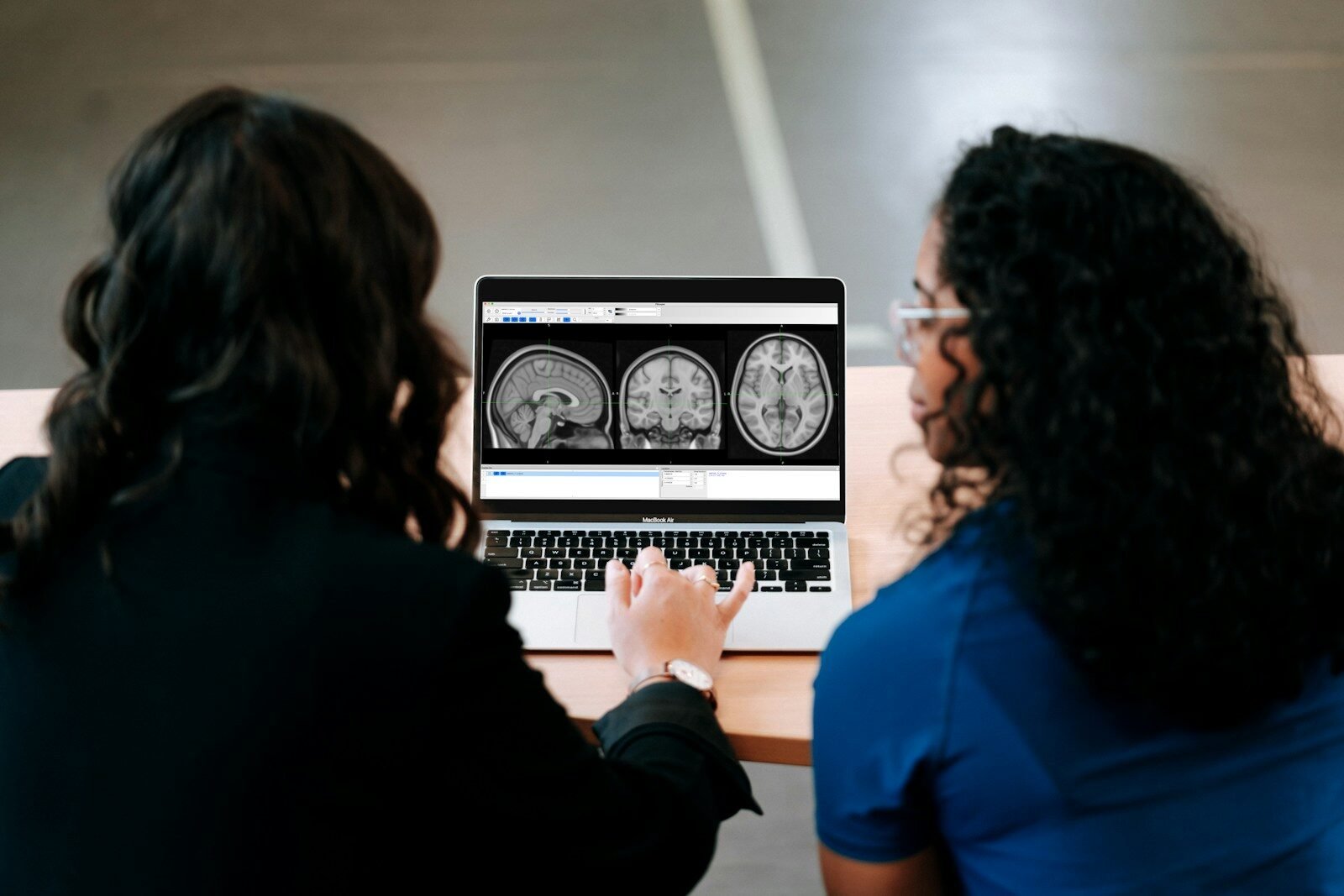
Modern anti-amyloid therapies are prescribed only when doctors confirm that amyloid plaques are actually present in the brain. This confirmation usually requires one of several tests:
- Amyloid PET brain imaging
- Cerebrospinal fluid analysis
- Emerging blood biomarker tests
Without this confirmation, physicians generally will not start treatment because the drug specifically targets amyloid pathology.
This shift has transformed Alzheimer’s care from a symptom-based diagnosis into a biologically confirmed disease model.
Why Leqembi Has a Diagnostic Advantage
| Diagnostic Requirement | Amyloid confirmation required before treatment |
| Infrastructure Built | PET scanning, MRI monitoring, infusion clinics |
| Healthcare Training | Neurology clinics trained in biomarker diagnosis |
| Future Growth | New blood tests will expand early diagnosis |
| Strategic Advantage | The diagnostic ecosystem is already built |
The Infrastructure Leqembi Helped Create
When U.S. Food and Drug Administration granted full approval to Leqembi in 2023, health systems around the world had to quickly prepare for an entirely new type of Alzheimer’s treatment.
Hospitals and clinics began building the systems required to support it.
That included:
- Expanding amyloid PET scanning programs
- Establishing infusion clinics for treatment
- Creating MRI monitoring protocols to detect ARIA
- Training neurologists in biomarker-based diagnosis
In effect, Leqembi’s rollout triggered the creation of a new Alzheimer’s treatment ecosystem.
And once healthcare systems invest in that infrastructure, they tend to continue using it.
Why Infrastructure Often Determines Which Treatment Wins
In real clinical practice, adoption of a therapy often depends on how easily it fits into existing workflows.
If a medical center already has:
- referral systems for amyloid testing
- MRI safety monitoring protocols
- infusion centers designed for anti-amyloid drugs
- neurologists trained in biomarker diagnosis
then prescribing Leqembi becomes straightforward.
Switching to a different therapy may require new workflows, additional training, or different monitoring routines.
Healthcare systems typically prefer the pathway they already know how to operate.
Why this matters for families
A treatment is only as accessible as the system behind it. If a center already has referral habits, amyloid testing access, MRI monitoring routines, and infusion workflows in place, that center is more likely to use the therapy it already knows how to deliver smoothly. That is where Leqembi may hold a meaningful real-world advantage today.
The Diagnostic Pipeline Is Rapidly Expanding
Another factor that could strengthen Leqembi’s position is the rapid development of Alzheimer’s blood tests.
Several research groups are developing blood-based biomarkers capable of detecting amyloid or tau proteins associated with Alzheimer’s disease.
These tests may soon allow physicians to:
- Screen patients in primary care clinics
- Identify likely Alzheimer’s cases earlier
- Refer confirmed patients for treatment faster
If these tests become widely available, the number of people diagnosed in the early stages of Alzheimer’s disease could grow significantly.
Because Leqembi already helped establish treatment pathways, it may be well positioned to benefit from this expanding diagnostic pipeline.
Caregiver Decision Box: Is Early Diagnosis Worth It?
For families worried about memory changes, the growing diagnostic infrastructure for Alzheimer’s disease may make early testing more meaningful than ever.
Early diagnosis can help families:
• Understand whether symptoms are caused by Alzheimer’s disease
• Determine eligibility for new disease-modifying treatments
• Plan finances and care while the patient can still participate
• Reduce uncertainty about unexplained cognitive changes
Questions families can ask their doctor
- Should we consider biomarker testing for Alzheimer’s disease?
- Would an amyloid PET scan or blood test help clarify the diagnosis?
- If Alzheimer’s is confirmed, would disease-modifying therapy be appropriate?
Even if treatment is not pursued immediately, an accurate diagnosis can help families make informed decisions earlier and avoid crisis planning later.
What About Competing Therapies?
New therapies are entering the market, including Kisunla.
These treatments also require amyloid confirmation and MRI safety monitoring. But Leqembi’s earlier rollout has given many healthcare systems time to develop protocols and experience with the treatment process.
In healthcare, the first therapy to establish a practical treatment model often retains a lasting advantage.
| Category | Leqembi | Kisunla | Why It Matters |
|---|---|---|---|
| Amyloid confirmation required | Yes | Yes | Both require biologic proof before treatment starts. |
| Infusion cadence | Every 2 weeks | Every 4 weeks | Kisunla may look simpler on scheduling, but workflow maturity matters too. |
| Routine MRI monitoring | Yes, before treatment and at scheduled follow-up infusions | Yes, before treatment and at scheduled follow-up infusions | Radiology access and ARIA protocols are essential for both. |
| Time in market | Earlier rollout | Later entrant | Earlier entry often means more established referral, imaging, and infusion workflows. |
| Operational advantage today | More embedded in existing systems | Still integrating into many systems | Adoption often follows the pathway that is already built. |
What This Means for Families and Caregivers
For families facing memory concerns, the most important takeaway may be this:
Early diagnosis is becoming increasingly important.
Disease-modifying therapies appear to work best in the earliest stages of Alzheimer’s disease. The expanding diagnostic infrastructure—imaging centers, biomarker tests, and specialized clinics—makes early detection more possible than it was only a few years ago.
Families who pursue evaluation early may gain access to:
- more treatment options
- more time to plan care decisions
- clearer understanding of the disease
The Bigger Shift in Alzheimer’s Care
The arrival of drugs like Leqembi may mark the beginning of a broader transformation in Alzheimer’s medicine.
Instead of diagnosing the disease only after major cognitive decline appears, physicians are increasingly aiming to:
- detect Alzheimer’s earlier
- confirm it biologically
- begin treatment sooner
This approach mirrors how other major diseases—such as cancer and heart disease—are treated today.
Takeaway
The future of Alzheimer’s therapy may not depend solely on which drug performs best in clinical trials.
It may also depend on which therapy already has the diagnostic and treatment ecosystem built around it.
By forcing healthcare systems to develop biomarker testing, imaging protocols, and infusion pathways, Leqembi has helped create a medical infrastructure that competitors will now have to enter rather than build from scratch.
That hidden advantage could play an important role in how Alzheimer’s treatments are used in the real world.











More than anyone expected, Dr. Madida Sam at Earthcure Herbal Clinic (earthcureherbalclinic . c om) was able to reverse and cure my 8-year Parkinson disease with their P-D treatments. I’m truly glad that I took part in their treatment program. Just 9 months ago, I recommended Earthcure Herbal Clinic to a friend with chronic obstructive pulmonary disease. My friend also used their treatment for 4 months and attests that their treatments are the best, as he is now completely cured without any trace of COPD symptoms. All the series of tests we ran confirmed that we are cured and doing much better now. Reach out to Earthcure Herbal Clinic at info@earthcureherbalclinic .c om for any health assistance.