For years, the medical community has celebrated the arrival of lecanemab (marketed as Leqembi) as a milestone in the fight against Alzheimer’s. While clinical trials proved the drug could slow cognitive decline and clear toxic amyloid-beta plaques, a fundamental question remained: how exactly does it trigger the brain to “clean itself”?
A groundbreaking study published in Nature Neuroscience has finally provided the answer. Researchers from VIB and KU Leuven have identified a specific part of the Leqembi molecule—the “Fc fragment”—as the essential key that unlocks the brain’s natural defense system. This discovery provides the first direct mechanistic explanation for how this class of drugs operates, moving the conversation from “what” it does to “how” it achieves its life-changing effects.
Leqembi – the bridge between the plaque and the cleanup crew
The human brain possesses its own dedicated immune system, led by specialized cells called microglia. In a healthy brain, these microglia cells act as a cleanup crew, patrolling the neural landscape to identify and remove cellular waste and debris. However, in patients with Alzheimer’s, this system breaks down.
Toxic amyloid-beta proteins begin to clump together, forming sticky plaques that damage neurons. While microglia typically swarm around these plaques, they often become “stalled” or ineffective. They are present at the site of the damage, but they lack the specific instructions or “fuel” required to actually consume and eliminate the toxic buildup. This is where Leqembi steps in, acting as a bridge between the plaque and the cleanup crew.
The Fc Fragment: The Hidden Engine Behind Leqembi’s Effect
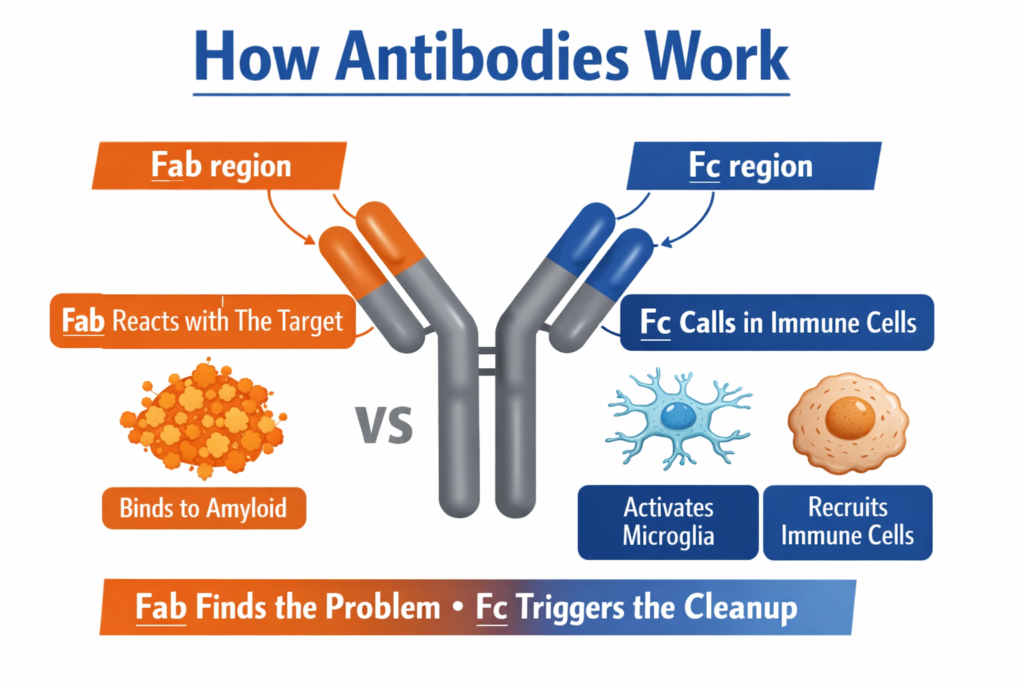
Most people think Alzheimer’s antibodies work simply by attaching to amyloid plaques. But that’s only half the story. The real cleanup job begins with a different part of the antibody called the Fc fragment.
Antibodies are Y-shaped proteins used by the immune system to recognize and remove harmful substances. Each antibody has two main functional regions.
| Antibody Region | What It Does |
|---|---|
| Fab region | Recognizes and binds to a specific target (such as amyloid in the brain) |
| Fc fragment | Signals immune cells to remove whatever the antibody has attached to |
The Fab region acts like a highly precise sensor. It identifies a target—in this case, the toxic forms of amyloid associated with Alzheimer’s disease.
But once the antibody attaches, nothing would happen without the Fc fragment.
The Fc fragment (Fragment crystallizable) is the “communication handle” that interacts with immune cells. It binds to Fc receptors on immune cells such as microglia, the brain’s resident cleanup cells.
When this interaction occurs, microglia receive a signal that something harmful has been tagged for removal.
How the Process Works in the Brain
Research suggests the mechanism of Leqembi follows a multi-step process:
- Leqembi antibodies bind to soluble amyloid aggregates in the brain through their Fab regions.
- The Fc fragment engages receptors on microglia.
- Microglia become activated and begin engulfing the tagged amyloid clusters.
- Over time, this leads to reduced amyloid plaque burden in the brain.
In other words, the antibody doesn’t simply stick to amyloid—it calls the brain’s immune system to clean it up.
Why This Mechanism Matters
Understanding the Fc fragment helps explain why some anti-amyloid antibodies work better than others.
Scientists now believe that successful Alzheimer’s antibodies must do two things well:
- Bind strongly to harmful amyloid species
- Activate microglia efficiently—but not excessively
Too little immune activation means amyloid remains in place. Too much activation may increase inflammation or side effects.
This delicate balance is one reason researchers carefully design the Fc region of therapeutic antibodies.
A New Insight Into Why Leqembi Works
Recent laboratory research suggests that the Fc fragment’s interaction with microglia may be essential for clearing amyloid aggregates. Without that immune activation step, simply binding amyloid may not be enough to remove it from the brain.
This insight helps explain why Leqembi—an antibody designed to target early amyloid aggregates while engaging microglia—has been able to demonstrate measurable reductions in amyloid and modest slowing of cognitive decline in clinical trials.
For caregivers and patients, the takeaway is simple: the drug is not just blocking amyloid—it is recruiting the brain’s own cleanup system to remove it.
Clinical Context and Safety Monitoring
While this mechanism explains why Leqembi is effective, it also highlights why medical supervision is vital. Because the drug works by activating an immune response, it can lead to inflammation. In some cases, this manifests as ARIA (Amyloid-Related Imaging Abnormalities), which involves temporary swelling or small spots of bleeding in the brain.
Understanding that the Fc fragment is the driver of this immune activity helps doctors understand why certain patients may react more strongly than others. It also explains why Leqembi is currently FDA-approved specifically for patients in the early stages of Alzheimer’s or those with Mild Cognitive Impairment (MCI). At these stages, the “cleanup crew” is still functional enough to be successfully reprogrammed.
What This Means for Families
For caregivers and families, this research offers a sense of clarity. It confirms that Leqembi is not merely a “chemical shield” but a tool that restores a vital biological process. It transforms the “stalled” immune cells of a loved one back into active protectors.
When discussing treatment options with a neurologist, families can now understand that the drug’s success depends on this cellular partnership. This also underscores the importance of the bi-weekly infusion schedule, which maintains a steady presence of these “anchors” to keep the cleanup crew working around the clock.
Looking Toward Next-Generation Therapies
The discovery that the Fc fragment is indispensable opens doors for future treatments. Scientists are now looking at ways to activate the microglial cleanup program without necessarily requiring a full antibody. This could lead to “next-generation” drugs that are even more efficient at clearing plaque with a lower risk of inflammatory side effects.
By defining the specific “cellular program” that drives plaque clearance, the VIB-KU Leuven team has provided a roadmap for the future of Alzheimer’s care. We are moving closer to a world where we don’t just slow the disease, but actively manage the brain’s environment to maintain health and cognitive function for longer.