NEWS VIDEO + ARTICLE:
17 Alzheimer’s supplement manufacturers received FDA warnings about 58 illegal below-standard products.
The U.S. Food and Drug Administration posted 12 warning letters and five online advisory letters issued to foreign and domestic companies that illegally sell more than 58 products, many that are sold as dietary supplements, which are unapproved new drugs and/or misbranded drugs that claim to prevent, treat or cure Alzheimer’s disease and a number of other serious diseases and health conditions.
These products, which are often sold on websites and social media platforms, have not been reviewed by the FDA and are not proven safe and effective to treat the diseases and health conditions they claim to treat. These products may be ineffective, unsafe and could prevent a person from seeking an appropriate diagnosis and treatment.
Protect Against Alzheimer’s Scams
“Science and evidence are the cornerstone of the FDA’s review process and are imperative to demonstrating medical benefit, especially when a product is marketed to treat serious and complex diseases like Alzheimer’s. Alzheimer’s is a challenging disease that, unfortunately, has no cure. Any products making unproven drug claims could mislead consumers to believe that such therapies exist and keep them from accessing therapies that are known to help support the symptoms of the disease, or worse as some fraudulent treatments can cause serious or even fatal injuries. Simply put, health fraud scams prey on vulnerable populations, waste money and often delay proper medical care – and we will continue to take action to protect patients and caregivers from misleading, unproven products,” said FDA Commissioner Scott Gottlieb, M.D.
Unapproved or Misbranded Alzheimer’s Drugs
The products cited in the warning and online advisory letters are unapproved new drugs and/or misbranded drugs that claim to prevent, treat or cure Alzheimer’s disease and a number of other serious diseases and health conditions, and have been sold in violation of the Federal Food, Drug, and Cosmetic Act. The products include a variety of product types, such as tablets, capsules and oils. The companies were asked to respond to the FDA within 15 days of receipt of the letters, stating how the violations outlined in the agency’s letters will be corrected. Failure to correct the violations promptly may result in legal action, including product seizure and/or injunction.
40 Alzheimer’s Warning Letters, 80 Products
As part of the FDA’s effort to protect consumers from Alzheimer’s disease health fraud, the FDA has issued more than 40 warning letters in the past to companies illegally marketing over 80 products making Alzheimer’s disease claims on websites, social media and in stores.
The FDA has also taken action in recent years against companies and dietary supplements making similar claims for the treatment of serious conditions such as cancer and opioid addiction. Although these companies may have stopped selling the products or making unproven claims, numerous unsafe and unapproved products continue to be sold directly to consumers due in part to the ease with which companies can move their marketing operations to new websites.
The FDA continues to encourage consumers to remain vigilant whether online or in a store in order to avoid purchasing products that claim to prevent, treat or cure diseases without any proof they will work. Health care professionals and consumers are also advised to report adverse reactions associated with these or similar products to the agency’s MedWatch program.
Supplement Warnings and Advisories
As part of its comprehensive efforts, … the FDA sent 12 warning letters and five online advisory letters to companies whose products, many of which are marketed as dietary supplements, are being illegally marketed as unapproved new drugs because the products bear unproven claims to prevent, treat or cure Alzheimer’s disease, as well as a number of other serious diseases and health conditions, including diabetes and cancer.
Products intended to treat Alzheimer’s disease must gain FDA approval before they are sold in order to help ensure they are safe and effective for their intended medical use.
Dietary supplements can, when substantiated, claim a number of potential benefits to consumer health, but they cannot claim to prevent, treat or cure diseases like Alzheimer’s. Such claims can harm patients by discouraging them from seeking FDA-approved medical products that have been demonstrated to be safe and effective for these medical conditions.
FDA Warning Lists & Letters
Lists & letters below video…
| Company Name (Click name for FDA Warning) |
Alzheimer’s Disease Product Name1 | Image2 (Click Image to Enlarge) |
|---|---|---|
| Blue Ridge Silver |
|

|
| BR Naturals |
|
 |
| DK Vitamins |
|
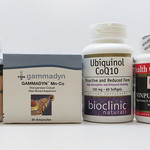 |
| Earth Turns, LLC |
|
 |
| Gold Crown Natural Products |
|
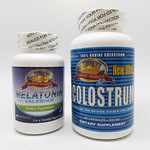
|
| Emmbros Overseas Lifestyle PVT LTD. |
|

|
| John Gray’s Mars Venus LLC |
|
 |
| Nutrition Coalition Inc | Alpha Lipoic Acid, CoQ10, Curcumin, Liva-Vite, Milk Thistle, Selenium, Vinpocetine, Vitamin B Complex, Vitamin C, Vitamin D3, and Vitamin E |  |
| Peak Nootropics LLC | Bacopa Monnieri, L-Theanine, Noopept, Piracetam, and Uridine | |
| Pure Nootropics |
|
 |
| Sovereign Laboratories, LLC | Colostrum-LD® and Curcumin-LD® | 
|
| TEK Naturals |
| 
|
| Online Advisory Letters | ||
| Alzheimer’s Corporation (ALZcorp) |
|
|
| Alzheimer’s Organization | Mind and Memory Support | |
| Dr. Fitt Intelligent Designs, LLC |
|

|
| Healthcare Products, LLC d/b/a/ The Alzheimer’s Store d/b/a MindCare Store |
|
|
| This Wonderful Life New Age Shop |
|
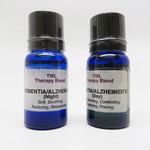 |
- The other products cited in the warning letters are not listed here.
- Product name linked to Flickr image when available.
REFERENCE:
SOURCES:
-
FDA: U.S. Food & Drug Administration
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency is also responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products. - FDA 2019 Warning Letters










It doesn't look like John Gray has changed his ways. He is still saying anything to make a buck. his videos are full of misinformation and he reccomends products based on "his own personal experiences". daughter hasn't fallen far from the tree it seems.
Perhaps if the FDA would ensure our food supply had adequate nurtrition and wasn't filled with pesticides, fungicides, heavy metals, GMOs, etc, we wouldn't be consuming so many supplements to try and fix all our health problems. They are so concerned we're being hurt by contaminated supplements, but yet they allow our food to be filled with toxic chemicals!!
We are able to control many things about our diet. Even if we couldn't not control anything, does that still justify taking contaminated or worthless supplements? Those supplements still make money for the frauds who sell them and the gullible folks who will believe anything in an infomercial. Worst of all, some of those supplements are harmful. (See the above list of people ending up in the ER because of the side effects.) Be smart about your diet AND about supplements.
Useless information if the supplements are not named.
I guess the lists at the links in the article above were not clear enough, so we went ahead and made up a really clear list, as you requested. Here it is, let me know what you think of it. Thanks.
FDA Warning List – Alzheimer's Supplements –
VIDEO + ARTICLE:
Also – the fact that Amazon's ads for Memory Supplements show up right in the middle of your article (multiple times) – in a format very similar to yours – makes it very difficult to tell what is your info and what is theirs…..
If dietary supplements are so dangerous how come there have been NO deaths from any of them reported by the Center of Disease Control for the last several years?
Well….. actually…..
"the Centers for Disease Control and Prevention and the Food and Drug Administration, tracked ER admissions at 63 hospitals throughout the United States from 2004 to 2013. Of the estimated annual 23,000 ER visits… suffering from heart palpitations, chest pain, choking or other problems after ingesting dietary supplements, 2,000 resulted in hospitalizations.
Read more details in plain English at the Washington Post article:
Dietary supplements send more than 23,000 people to the ER each year
The article should name the products.. otherwise, it's useless.
There are links in the article itself as well as in the REFERENCE section at the end of the article to the actual warning and advisory letters that I believe DO name the companies and products. Hope this helps.
I agree with Alice Liu. After reading everything above and then find out they aren't listed is a waste of time!!!
I guess the lists at the links were not clear enough, so we went ahead and made up a really clear list, as you requested. Here it is, let me know what you think of it. Thanks.
FDA Warning List – Alzheimer's Supplements
VIDEO + ARTICLE:
I'm still unclear – was the issue about the production / quality of these particular supplements? Or the claims that were made? Many of these are important supplements in appropriate situations (D3; Omega 3s, etc.) – maybe just the wrong manufacturers????