Leqembi (lecanemab) is one of the first Alzheimer’s treatments designed to slow the disease itself — not just temporarily improve symptoms. Because of both its potential benefits and its risks, doctors follow very strict eligibility criteria. Many families assume a diagnosis of Alzheimer’s automatically means someone can receive it. In reality, only a subset of patients qualify.
This guide walks step-by-step through how doctors determine eligibility. (Continued below video…)
Step 1: The Stage of Alzheimer’s Matters Most
Leqembi is approved only for early Alzheimer’s disease, specifically:
- Mild Cognitive Impairment (MCI) due to Alzheimer’s
- Mild dementia due to Alzheimer’s
It is not approved for:
- Moderate Alzheimer’s
- Severe Alzheimer’s
- Advanced dementia from any cause
Why?
The drug removes amyloid plaques. Once extensive brain injury has already occurred, removing plaques no longer meaningfully changes the course of the disease.
Practical rule:
If the person still lives mostly independently but struggles with memory — possible candidate.
If they need daily supervision for basic tasks — usually too late.
Step 2: You Must Prove Amyloid Is Present
A clinical diagnosis alone is insufficient.
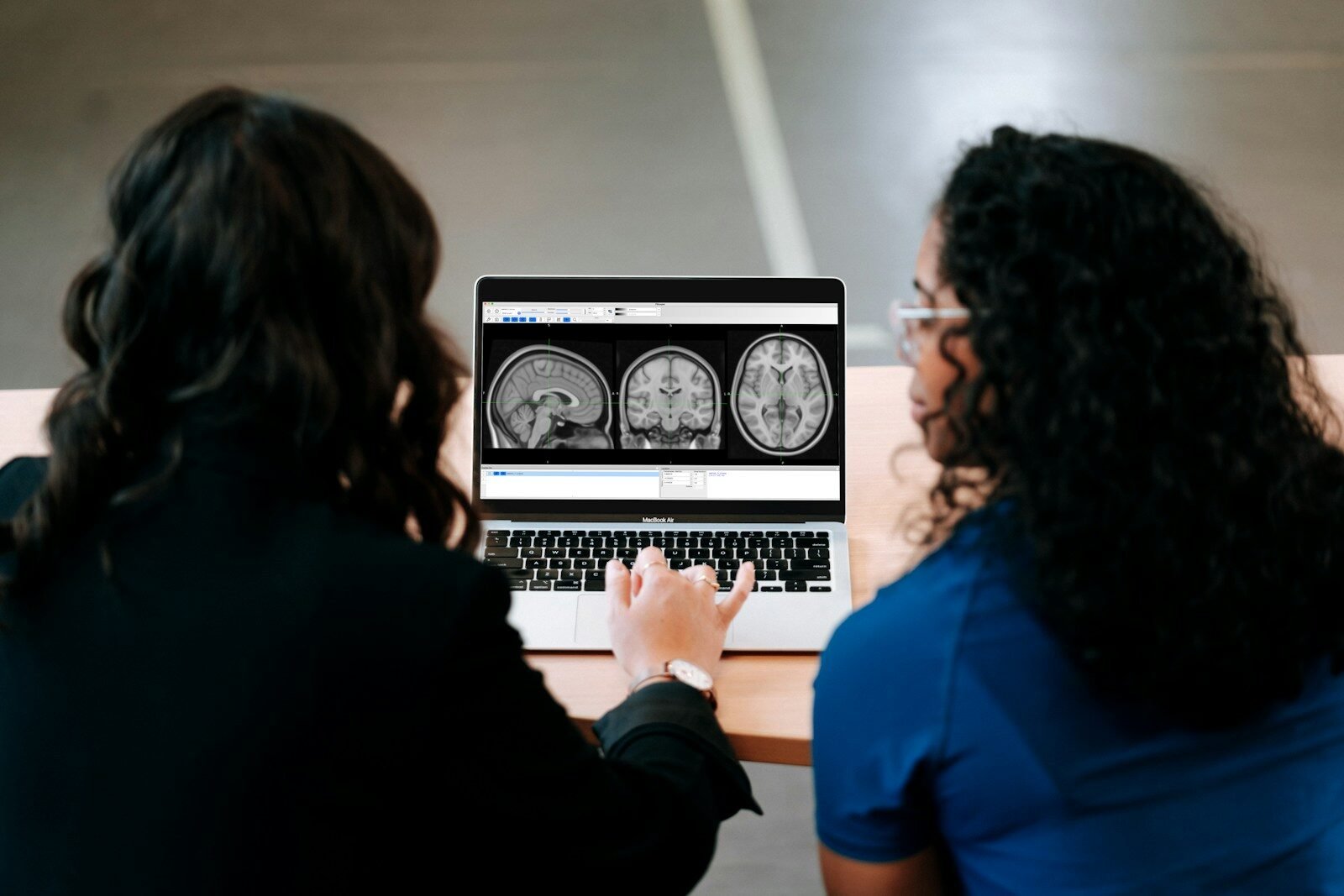
Before prescribing Leqembi, doctors must confirm amyloid buildup in the brain.
This is done using one of two tests:
PET Amyloid Scan
A specialized brain scan that visually shows amyloid plaques.
Lumbar Puncture (Spinal Tap)
Measures amyloid and tau proteins in spinal fluid.
If amyloid is not confirmed → Leqembi cannot be given.
This is important because many conditions mimic Alzheimer’s:
- Vascular cognitive impairment
- Lewy body dementia
- Depression-related cognitive decline
- Medication side effects
Treating the wrong disease exposes patients to risk without benefit.
Step 3: MRI Screening for Brain Bleeding Risk
Leqembi can cause a side effect called ARIA (Amyloid-Related Imaging Abnormalities) — small brain swelling or bleeding visible on MRI.
Most cases are mild, but some can be serious.
Before treatment, doctors order an MRI to check for:
Disqualifying findings include:
- Multiple microbleeds
- Prior brain hemorrhage
- Certain vascular abnormalities
- Extensive small-vessel disease
If risk is high → treatment is unsafe.
Patients must also undergo repeated MRIs during therapy (typically at months 3, 5, and 7).
Step 4: Genetic Risk Discussion (APOE4 Testing)
Patients are usually offered a blood test for the APOE4 gene.
People with two copies (homozygous APOE4) have a much higher risk of ARIA complications.
They are not automatically excluded, but doctors often recommend against treatment or proceed cautiously.
This is a shared decision — not a strict rule — but many centers require genetic counseling before proceeding.
Step 5: Medication and Medical History Review
Certain medications and conditions make Leqembi dangerous.
Often Excluded
- Blood thinners (especially warfarin)
- Recent stroke
- Seizure disorders
- Uncontrolled hypertension
- Significant bleeding disorders
Some newer anticoagulants may be allowed depending on the center, but policies vary.
Step 6: Functional and Support Requirements
Because Leqembi requires biweekly IV infusions and monitoring, practical eligibility matters too.
Patients must have:
- A reliable caregiver
- Ability to attend infusion appointments every 2 weeks
- Ability to report symptoms quickly
- Access to MRI monitoring
This is not just convenience — early recognition of side effects prevents severe complications.
Step 7: Understanding What the Drug Actually Does
Leqembi does not reverse Alzheimer’s and does not stop decline completely.
In studies, it slowed progression by about 27% over 18 months — meaning the disease advances more slowly, not that symptoms disappear.
Patients who expect improvement are often disappointed.
Patients who understand the goal is preserving current abilities longer tend to benefit most.
Doctors therefore assess whether the patient and family understand the realistic outcome before prescribing.
Quick Eligibility Checklist
A likely candidate usually meets all of the following:
- Early Alzheimer’s symptoms only
- Amyloid confirmed by PET or spinal tap
- MRI shows low bleeding risk
- Acceptable medication profile
- Caregiver support available
- Willing to undergo repeated monitoring
- Understands the goal is slowing decline
Failing any major category usually means the risks outweigh benefits.
The Most Common Reason Patients Don’t Qualify
Not late stage — wrong diagnosis.
Large numbers of people labeled “Alzheimer’s” actually have mixed dementia or vascular disease. Leqembi only works on amyloid-driven Alzheimer’s.
This is why the biomarker step is mandatory.
Bottom Line
Leqembi eligibility is less about severity of symptoms and more about biology and safety risk.
Think of it this way:
The drug is appropriate for the right disease, at the right time, in the right brain.
Families often view testing as a barrier. In reality, the testing protects patients from receiving a powerful therapy that could harm them without benefit.
If you’re considering Leqembi, the best first step is not asking “Can we get the drug?”
It is asking your doctor:
“Can we confirm whether this is early amyloid Alzheimer’s?”
That single question determines everything that follows.
More Information for Caregivers
Ask your doctor these 5 questions before pursuing Leqembi:
- Has Alzheimer’s been confirmed with an amyloid PET scan or spinal tap — or only suspected?
- What stage is the disease right now: MCI, mild, or moderate?
- Is an MRI safe for this patient, and what is their ARIA risk?
- Are current medications (especially blood thinners) a problem?
- What benefit should we realistically expect in daily life?
Signs Someone May Be a Good Candidate
- Still manages most daily activities independently
- Repeats questions or forgets recent conversations
- Gets lost in complex tasks but functions at home
- Symptoms began within the past few years
- A caregiver can reliably attend biweekly infusions
Signs Treatment May Not Help
- Needs supervision most of the day
- Cannot manage medications or finances
- Personality or behavior changes dominate symptoms
- Diagnosis uncertain or mixed dementia suspected
- MRI shows significant vascular disease
What Families Often Misunderstand
Leqembi does not improve memory for most patients.
Its purpose is to preserve current abilities longer — delaying decline rather than reversing it.
The best outcomes occur when expectations are realistic and treatment begins early.
Action Step:
If you are considering Leqembi, request a referral to a memory clinic and ask specifically for biomarker confirmation of Alzheimer’s disease.
That single step determines whether treatment is appropriate.