The Headline That Shook the Alzheimer’s World
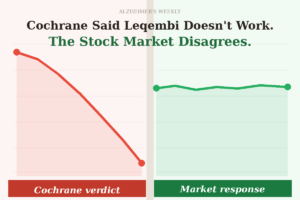
Last week, one of Britain’s most influential newspapers declared that Alzheimer’s “wonder drugs do not work.”
That was the headline in The Daily Telegraph — a 170-year-old broadsheet with a global readership in the millions, and one of the most authoritative and widely-cited voices in British and international journalism. This was not a fringe outlet or a clickbait blog. When the Telegraph runs a headline like that, it lands.
For families already navigating the exhausting uncertainty of this disease, it landed very hard.
But there is a significant problem with the study behind that headline — one that many of the world’s leading Alzheimer’s researchers are calling out publicly and directly. The analysis, they say, mixed drugs that never worked with drugs that do, and then used the combined average to condemn them all.
What the Cochrane Review Actually Found
The review was published by the Cochrane Collaboration, a global nonprofit widely respected for rigorous evidence synthesis. It pooled data from 17 clinical trials testing seven different drugs designed to clear amyloid plaques from the brain — a class known as anti-amyloid therapies.
Its conclusion: these drugs do not improve cognitive function, do not slow decline, and carry real risks of brain swelling and bleeding.
The two most prominent drugs in the review were:
- Leqembi (lecanemab) — FDA-approved in 2023
- Kisunla (donanemab) — FDA-approved in 2024
Both were hailed as genuine milestones when they reached the market — the first treatments to show they could actually modify the course of Alzheimer’s disease, not just manage symptoms.
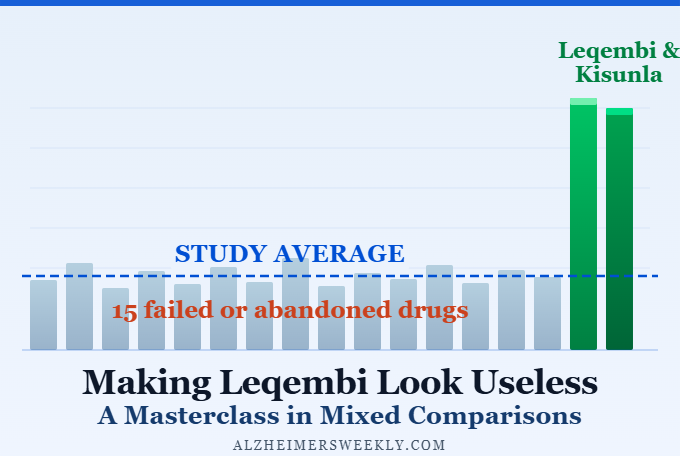
The Core Problem: 15 Failures, 2 Successes — Averaged Together
Here is what makes many scientists angry about this review.
Of the 17 trials analyzed, only one examined Leqembi and one examined Kisunla. The remaining 15 trials studied drugs that failed — drugs that were abandoned and never approved precisely because they didn’t work.
Prof. Steve Macfarlane of Dementia Support Australia made the point plainly: “I really don’t think it’s a fair comparison to look at 15 failed trials and two trials that have clearly been successful and led to approval of the drugs by the FDA, and say that on average these drugs don’t work.”
He also noted something striking: the forest plots within the Cochrane review itself — the internal charts showing each drug’s individual results — show a clear benefit for both Leqembi and Kisunla. The effect size is modest. But it is visible, unambiguous, and consistent.
“It’s not a dramatic, world-changing benefit,” Macfarlane said, “but it’s a clear and obvious and undeniable benefit.”
What Leading Scientists Are Saying
The pushback has been swift and pointed — and it’s coming from independent researchers with no financial stake in the drugs.
Prof. Bart De Strooper of the UK Dementia Research Institute put it directly: “This review does not clarify the evidence, it blurs it. By mixing failed drugs with the only antibodies that have actually changed clinical practice, it turns therapeutic progress into statistical noise.”
Prof. Tara Spires-Jones of the University of Edinburgh noted that the conclusions were weakened by including five drugs that never passed their clinical trials and are not available to patients. She pointed to multiple regulators — including the European Medicines Agency and the UK’s Medicines and Healthcare products Regulatory Agency — whose careful, independent reviews support the use of both Leqembi and Kisunla to slow decline in early Alzheimer’s disease.
Prof. Dag Aarsland of King’s College London said that while the class-level analysis is open to question, he considered the conclusions supporting the individual drug approvals to remain valid.
How Much Benefit Are We Actually Talking About?
This is the honest part — and it matters for caregivers especially.
Clinical trial data for Leqembi and Kisunla suggests they may slow the progression of cognitive decline by roughly 25–35%. Patients who begin treatment at an earlier stage of illness appear to get the most benefit. Data now extends to four years, suggesting that benefit doesn’t simply fade after the 18-month window that the Cochrane review focused on.
That is progress. It is not a cure, and it should not be described as one.
Dr. Susan Kohlhaas of Alzheimer’s Research UK raised an important point: there is no universally agreed definition of what “clinically meaningful” even means in this context. “We regularly hear from families affected by dementia that even a delay of several months in decline could provide valuable, meaningful time with loved ones.”
That framing matters enormously to the people actually living with this disease.
What This Means for Caregivers
If someone in your family is in the early stages of Alzheimer’s disease, here is what to take away:
- The Cochrane review does not prove these drugs are useless — it proves that averaging failed drugs with successful ones produces a misleading result.
- Leqembi and Kisunla remain FDA-approved treatments. Their approval was based on rigorous, independent clinical trials reviewed by multiple regulatory bodies.
- The benefit is modest and is most meaningful for people in the earliest stages of the disease.
- Speak with a neurologist or memory specialist before drawing any conclusions from this study about your family member’s treatment options.
This is not the moment to give up on a treatment plan. It is the moment to ask better questions.
The Field Is Honest About Its Limits — And That’s a Good Sign
Science works through challenge and counter-challenge. The Cochrane review raised real questions. The responses from independent researchers have been substantive, not defensive.
What both sides agree on: the benefit of current drugs is modest, treatment works best early, and the field needs more rigorous and independent long-term data.
For caregivers, that means one thing above all: the window of opportunity for early intervention matters. If you have concerns about a loved one’s memory or cognition, pursuing evaluation sooner rather than later remains the right course of action.
Alzheimer’s Weekly will continue to follow this story as expert analysis of the Cochrane review evolves.
If this article helped clarify the picture, share it with someone who needs to see it — or leave a comment below with your questions.