The Daily Telegraph — one of the most trusted newspapers in the English-speaking world — just ran the headline:
“Alzheimer’s Wonder Drugs Do Not Work.”
For anyone who had built hope around Leqembi or Kisunla, it was a gut punch.
And then something unexpected happened. Experts — whose entire job is to react to exactly this kind of news — barely noticed.
Indifference
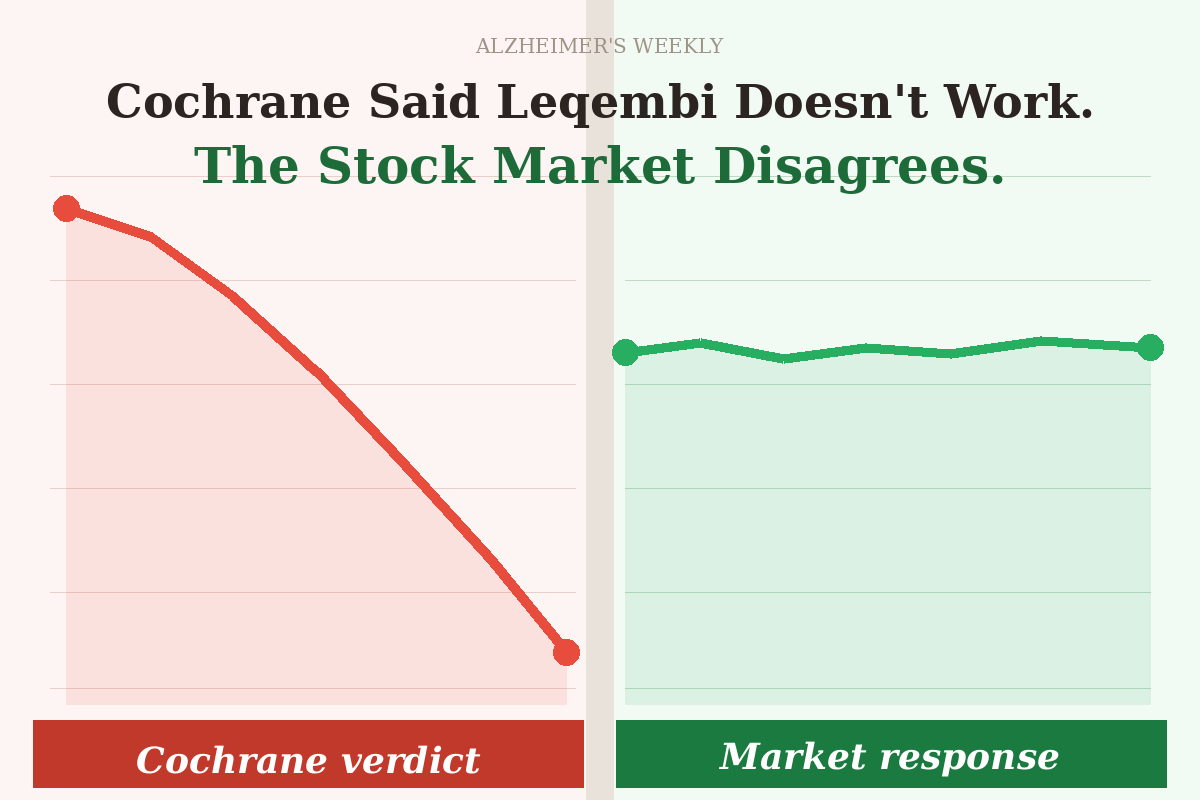
When the Cochrane Collaboration — one of the most respected evidence-review organisations in the world — published a sweeping analysis concluding that Leqembi and similar Alzheimer’s drugs provide no meaningful benefit to patients, experts watching the drug responded with something close to indifference.
For the millions of families living with Alzheimer’s disease who woke up to alarming headlines that morning, that reaction is worth understanding. Because the non-response by experts – from scientists to investors -may be the most honest verdict available on what the Cochrane study actually proved.
The reaction of scientists and researchers are covered in the next two parts of this series, as they require a little technical background. There are links at the end of this article, as well as here:
- Cochrane’s Leqembi Ruling Used Dr. Andrews’ Bar “Inappropriately”
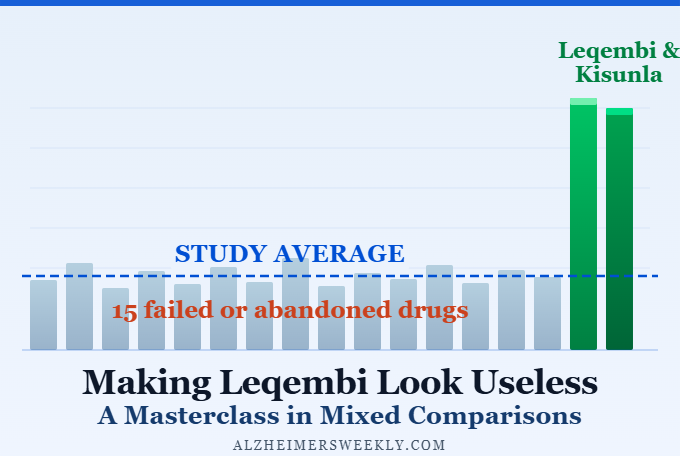
- Making Leqembi Look Useless: A Masterclass in Mixed Comparisons
What follows is how the markets responded, as everyone understands that all the markets care about is whether or not a drug is worthwhile.
The People Who Read the Small Print
Here’s the key: institutional investors in pharmaceutical stocks employ people specifically to read clinical trial data. They were already well aware that Leqembi’s benefit is modest — slowing cognitive decline by around 25–35% in early-stage patients, rather than halting or reversing it. That was never a secret. The original trial data made it plain.
What the Cochrane review didn’t tell the market was anything it didn’t already know. And crucially, a significant methodological concern — averaging Leqembi’s results together with five drugs that failed their clinical trials and never reached patients — was visible immediately to anyone who read past the abstract.
The numbers tell a clear story:
| Company | Role | Market move |
|---|---|---|
| Biogen | Co-markets Leqembi | −2% |
| Eisai | Co-markets Leqembi | −0.2% |
| Eli Lilly | Makes rival drug Kisunla | +slight gain |
These numbers simply reflect a collective judgment that the study did not change the underlying reality.
A Positive Signal That Got Buried
There was another piece of context the market was weighing that barely made the news that day. The same week the Cochrane review published, the UK’s National Institute for Health and Care Excellence quietly confirmed it was reassessing its previous rejection of Leqembi and Kisunla for NHS use — under new cost-effectiveness thresholds agreed as part of a US-UK trade framework.
In other words, the regulatory direction of travel in Britain was moving toward the drug at the very moment the headlines were saying the opposite. Investors noticed. Most patients and their families didn’t.
What This Means for Caregivers
We want to be clear about what we are and aren’t saying. Leqembi is not a cure. Even its strongest advocates consistently describe it as a modest first step — a drug that buys time, slows the clock a little, and proves that slowing Alzheimer’s is biologically possible. That is genuinely meaningful after decades of failure, but it is not the end of the story.
What the Cochrane review did not do — whatever its headlines suggested — is prove that Leqembi doesn’t work, or that regulators in the US, UK, Europe, Japan and 49 other countries who approved it were wrong to do so.
Practical steps for families:
-
- If your loved one is in the early stages of Alzheimer’s, ask their neurologist specifically whether they qualify for Leqembi or Kisunla — eligibility is narrow, and most patients won’t meet the criteria.
-
- Don’t let alarming headlines drive a decision either way. Ask your doctor: “Has anything about this drug changed since the Cochrane review?”
-
- If you’re in the UK, it’s worth monitoring the NICE reassessment. If thresholds change, NHS access could follow within months.
-
- Keep in mind that Leqembi is one piece of a care strategy, not a replacement for other interventions — exercise, sleep, cognitive engagement, and caregiver support all remain evidence-backed priorities.
The experts said the drug still has merit. And then, quietly and without fanfare, so did the market.
If you want to understand why the study itself was flawed — not just how the world reacted to it — Part 2 of this series, Making Leqembi Look Useless: A Masterclass in Mixed Comparisons, breaks down exactly where the methodology went wrong. Also, check out Part 3: “Cochrane’s Leqembi Ruling Used Dr. Andrews’ Bar “Inappropriately”.
If this article helped you cut through the noise, use the SHARE BUTTONS below to share it with someone who’s navigating the same confusion — or leave a comment below with questions you’d like us to tackle next.