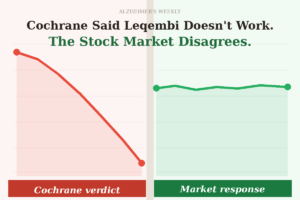
The Food and Drug Administration (FDA) has granted traditional approval for generic lecanemab (brand name Leqembi®). The new treatment promises to slow the decline of memory and thinking in some Alzheimer’s patients.
Dr. Kavita Patel joined Alicia Menendez on American Voices to discuss the drug’s benefits, risks and what it means for caregivers.
Lecanemab is a monoclonal antibody that has shown promise in removing amyloid plaques from the brain, according to phase 3 trial results.
Amazon Books & Videos
about Alzheimer’s and Dementia:
FREE Newsletter:
See Dr. Ronald Petersen, a neurologist and director of the Mayo Clinic Alzheimer’s Disease Research Center, describe Lecanemab’s therapeutic ability to slow Alzheimer’s by removing amyloid plaques. These plaques are one of the defining features of Alzheimer’s disease.
SOURCES: